卫生署规定浓度限量为 5%[1]
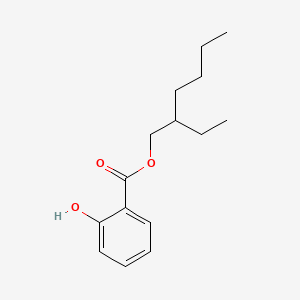
This ingredient can be used as UVB absorber. It also has a floral odor that might be used as a fragrance I gusess?
Ethylhexyl salicylate, or octyl salicylate (Octisalate), is an organic compound used as an ingredient in sunscreens and cosmetics to absorb UVB (ultraviolet) rays from the sun. It is an ester formed by the condensation of a salicylic acid with 2-ethylhexanol. C15H22O3. https://pubchem.ncbi.nlm.nih.gov/compound/2-ethylhexyl_salicylate
A colorless to light yellowish oily liquid that works as a UVB (280-320nm) sunscreen filter with a peak absorbance at 306 nm. It's not a strong filter in itself, it's always used in combination with other sunscreen agents to further enhance the SPF and to solubilize other solid UV filters.
It has a good safety profile and is allowed to be used at a max concentration of 5% both in the US and in Europe (10% is allowed in Japan).
https://incidecoder.com/ingredients/ethylhexyl-salicylate
Octisalate, an organic UV filter, readily absorbs through the skin at levels 10 times more than 0.5 nanograms per milliter, the FDA’s cutoff for systemic exposure. This cutoff is the maximum concentration that may be found in blood before there are potential safety concerns. The FDA has requested additional safety testing when a sunscreen is absorbed above this level (Walters 1997, Matta 2020).
The FDA 2019 proposed update suggests there is insufficient data to determine whether octisalate can be classified as safe and effective to use in sunscreens (FDA 2019). A case report showed that the chemical has been linked to allergic contact dermatitis (Singh 2007). Analysis of high throughput screening assays by the Environmental Protection Agency suggests octisalate may have endocrine effects, weakly binding to the estrogen receptor.
https://www.ewg.org/sunscreen/report/the-trouble-with-sunscreen-chemicals/
